The Kazak lab studies host energy metabolism in physiology and disease. We combine biochemical and genetic tools with mouse genetics to define key pathways that contribute to energy balance, metabolic syndrome and chronic disorders associated with metabolic dysfunction. These include obesity, diabetes and cancer.
Some ongoing projects:
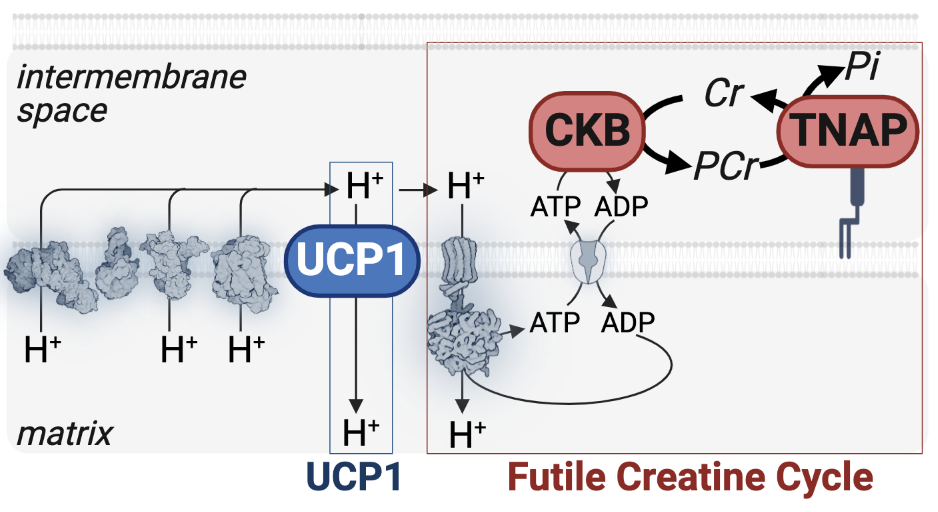
1. UCP1-independent thermogenesis. Creatine drives energy expenditure in adipose tissue by stimulating a futile creatine cycle [i], and this process can counter obesity and glucose dysregulation in pre-clinical models [ii], [iii], and [iv]. Our experimental evidence points to a two-enzyme system that uses mitochondrial-derived ADP phosphorylation to support cycling between creatine and phosphocreatine. We have defined creatine kinase, brain-type (CKB) as a key protein that regulates the first step of the futile creatine cycle [iv]. CKB, known primarily as a non-mitochondrial enzyme, is surprisingly the major mitochondrial creatine kinase isoenzyme in brown adipocytes. We find that due to a unique internal mitochondrial targeting signal, CKB traffics to mitochondria. Once translocated to these organelles, CKB triggers the initial reaction of a creatine phosphorylation cycle and simultaneously liberates mitochondrial ADP to increase the rate of mitochondrial respiration. We have constructed a new mouse model, wherein a Flag epitope tag has been inserted at the carboxy-terminus of the endogenous CKb locus (Ckb.Flag mice). We are combining Ckb.Flag mice with quantitative TMT-based proteomics to identify covalent modifications on CKB in response to acute thermogenic triggers. This work may lead to the identification of acute regulation of the futile creatine cycle.
[i] Kazak L et al., (2015) Cell ; [ii] Kazak L et al., (2017) Cell Metabolism ; [iii] Kazak L et al., (2019) Nature Metabolism ; [iv] Rahbani JF et al., (2021) Nature ; [v] Rahbani JF et al., (2022) Nature Metabolism
2. Determine the role of thermogenic effectors on combating obesity. The conventional view is that uncoupling protein 1 (UCP1) is the sole effector of thermogenesis within adipocytes, which is largely based on work from germline Ucp1 knockout (Ucp1-/-) mice. While the Ucp1-/- mouse model has advanced our understanding of adipocyte thermogenesis, there are limitations to its use because it acquires secondary changes beyond UCP1 loss [i], such as reduced expression of proteins regulating fuel liberation and combustion, which are changes that would attenuate the capacity of any thermogenic pathway. Hence, the physiological relevance of UCP1-dependent and -independent factors within adipose tissue in vivo remains unresolved. To continue our work on elucidating the role of adipocyte thermogenesis (UCP1 and beyond) in combating obesity and metabolic disease, we are generating new genetically-engineered mouse models where we can inactivate thermogenic genes selectively in fat, in an inducible manner.
[i] Kazak L et al., (2017) PNAS
3. Obesity-driven cancer progression. We are using functional genomic screens in vivo to understand the mechanisms underlying obesity-accelerated breast cancer progression. Many other cancer types are supported in an obese environment, so these screens can be applied to other cancer types as well.
[i] Kazak L and Cohen P (2020) Nature Reviews Endocrinology
Sources of Funding





